Getting a Structure from Multiple Tomograms of HIV Capsids (walkthrough)
In this walkthrough you will get familiar with managing multiple tomograms through the Dynamo catalogue and developing strategies for alignment projects to reach a reasonable structure from a set of tomograms. You will make use of the knowledge that you acquired during the workshop and apply it to a more realistic case. It is recommended to get familiar with at the advanced starters guide and the walkthrough for lattices on vesicles before starting this walkthrough.
Contents
Data
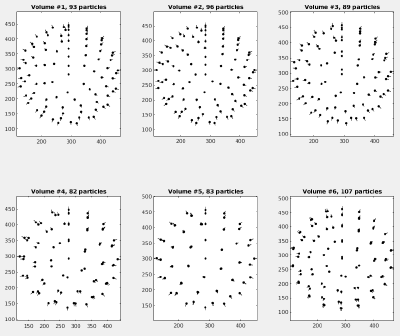
We prepared a set of 6 tomograms, each containing one Immature HIV-1 virus like particle (VLP) with a layer formed by a lattice of capsid proteins. The capsid proteins have a C6 symmetry, a diameter of roughly 15nm and a molecular weight of about 150kDa. The pixelsize of the tomograms is 2.7 angstrom. You can find the tomograms in:
~/data/tutorial_VLPs
The tomograms are part of the data that was used in the publication An atomic model of HIV-1 capsid-SP1 reveals structures regulating assembly and maturation by Schur FK, Obr M, Hagen WJ, Wan W, Jakobi AJ, Kirkpatrick JM, Sachse C, Kräusslich HG and Briggs JA. (2016). The full dataset can be found on the Electron Microscopy Public Image Archive (EMPIAR) under EMPIAR-10164. The 6 tomograms were extracted from the tilt series TS_03 and TS_43.
Goal
From the data given you should be able to get a final average in which you start seeing hints of alpha helices, i.e., it should be possible to reach a resolution close to roughly 10 angstroms.
Walkthrough
Set up a catalogue
Create a new catalogue
dcm -create catVLP
and open it in the catalogue manager
dcm catVLP
You can add each volume manually to the catalogue under Catalogue -> Browse for new volume or you can make use of a volume file and add all tomograms at once under Catalogue -> Input list of tomograms -> Browse for text file (.vll file). The volume file should be created beforehand by opening a blank text file named for example VLPtomograms.vll and copy the paths of all tomograms into it:
vlp_1.mrc vlp_2.mrc vlp_3.mrc vlp_4.mrc vlp_5.mrc vlp_6.mrc
In case you imported the tomograms using a volume file you have to click the button list volumes to refresh the tomogram list in the catalogue manager. Now you should see the 6 tomograms in the catalogue manager. Prebin the tomograms once (1x) under Catalogue -> Create binned versions -> with factor 1. You can now look at single tomograms in tomoslice by selecting a tomogram of choice in the catalogue manager and then go under View volume -> Load full prebinned Volume [if available] -> open in tomoslice. If needed, adjust the lowpass level and contrast in tomoslice to improve the depiction.
Annotate tomograms
To know where we want to later extract the subvolumes we have to first annotate the surfaces of the VLPs inside the tomograms. We do this by opening a tomogram inside tomoslice where a so called dipole Set model is defined. How to do that is described in detail in the section DipoleSet models, Corrections during picking and Corrections after picking of the walkthrough for lattices on vesicles. A new dipole set model has to be made for each tomogram. Save each model before closing tomoslice and when opening a new tomogram in tomoslice select Delete from memory if asked. In case you see markers of the previously defined dipole set inside the newly opened tomogram make sure the model pool is empty and click on reset scene before defining the new dipole set. Once you went through all tomograms click on the button list volumes inside the catalogue manager to refresh the list of tomograms. In the column model files of the tomogram list of the catalogue manager you should now see that you have one defined model per tomogram.
Define crop points
To extract subvolumes (crop particles) we need to first define the crop points on the surface of the VLPs using the previously defined models. The crop points are then stored in a table. This is all done with the following script which is an extended version of the one found in the walkthrough for lattices on vesicles.
% read dipole from catalogue into workspace
dcmodels catVLP -tc dipoleSet -ws o -gm 1
c=1; % counter for table
for tomo = 1:6 % loop over tomograms
ds = o.models{tomo}; % read models
NDipoles = length(ds.dipoles);
for i=1:NDipoles % loop over models (in our case just one per tomogram)
v = dmodels.vesicle(); % create empty vesicle model
v.center = ds.dipoles{i}.center; % add center from dipole to vesicle model
v.radius = norm( ds.dipoles{i}.north - ds.dipoles{i}.center); % add radius
v.separation = 60; % separation of crop points (in px)
v.crop_distance_from_surface =0;
v.updateCrop(); % update vesicle model
tv{c} = v.grepTable(); % create crop table from using vesicle model
tv{c}(:,22) = i; % add model number to table
tv{c}(:,20) = tomo; % add tomogram number to table
c=c+1;
end
end
In the standalone version you need to proceed as follows:
1.) Open a text file named commands.m and copy the following into the file (avoid commenting with % or #):
o =dcmodels('catVLP','type','dipoleSet','gm',1);
c=1;
for tomo = 1:6 % loop over tomograms
ds = o.models{tomo};
NDipoles = length(ds.dipoles);
for i=1:NDipoles
v = dmodels.vesicle();
v.center = ds.dipoles{i}.center;
v.radius = norm( ds.dipoles{i}.north - ds.dipoles{i}.center);
v.separation = 60;
v.crop_distance_from_surface =0;
v.updateCrop();
tv{c} = v.grepTable();
tv{c}(:,22) = i;
tv{c}(:,20) = tomo;
c=c+1;
disp(sprintf('Dipole %d will provide %d crop points',i,size(v.crop_points,2)));
end
end
dwrite(tv,'cellOfTables.mat');
2.) Execute the text file in a system shell or in the Dynamo console:
dynamo commands.m
3.) When you are back in the Dynamo console, you can load the results from the script into the workspace with
tv = dread('cellOfTables.mat');
Type whos to display the actual matlab workspace and to check if the variable tv was loaded correctly.
Write table with crop points
The tables beloning to each dipole model are stored in the cell array tv. To create one table containing all points we merge all tables into one with the command
tAll = dynamo_table_merge(tv,'linear_tags',1);
and visualize the table to see the crop points and their orientations
dtplot(tAll,'pf','oriented_positions'); axis equal
You should get something like:
Extract subvolumes
Before extracting the subvolumes (cropping particles) using the previously generated table we have to define a so called tomogram table map file which links the tomogram identification number in column 20 of the previously created table to an actual tomogram on the disk. Create a file named VLPtomograms.doc and copy the tomogram numbers and full paths of the tomograms into it:
1 vlp_1.mrc 2 vlp_2.mrc 3 vlp_3.mrc 4 vlp_4.mrc 5 vlp_5.mrc 6 vlp_6.mrc
Define a destination for the cropped particles:
targetFolder = './particlesSize128';
Now we can crop the particles with sidelength 128 pixels from the tomograms using the table previously defined.
dtcrop('VLPtomograms.doc',tAll,targetFolder,128);
The cropping on one core should not take longer than 5 minutes. Once done, you can look at the cropped particles with
dslices(targetFolder,'projy','*','t',finalTable,'align',1,'tag',1:10:500,'labels','on');
We create a plain average of all cropped particles which will be used later in the alignment project and can help us to asses the quality of the cropped particles. First we read the table of the cropped particles:
finalTable = dread([targetFolder '/crop.tbl']);
Then we form the average
oa = daverage(targetFolder,'t',finalTable,'fc',1);
and inspect the average with
dview(oa.average)
You should see a faint and noisy curvature of a thick membrane-like structure in the x and y direction. Save it with
dwrite(oa.average,[targetFolder '/template.em']);
First alignment project
Now we create our first alignment project through the command line in a similar way as described in detail in the walkthrough for lattices on vesicles. First we define a project name
pr = 'aliVLP';
Then we create the alignment project with the particles, template and table that we defined before. The masks are set to default.
dcp.new(pr,'d',targetFolder,'t',[targetFolder '/crop.tbl'],'template',[targetFolder '/template.em'],'masks','default','show',0);
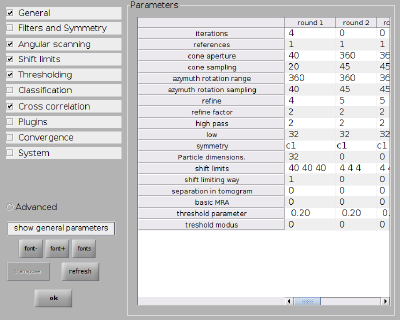
Next we set up the numerical parameters for the project (type dvhelp in the console for an overview of all parameters):
dvput(pr,'ite_r1',4); dvput(pr,'dim_r1',32); dvput(pr,'cr_r1',40); dvput(pr,'cs_r1',20); dvput(pr,'ir_r1',360); dvput(pr,'is_r1',40); dvput(pr,'rf_r1',4); dvput(pr,'rff_r1',2); dvput(pr,'lim_r1',[40,40,40]); dvput(pr,'limm_r1',1); dvput(pr,'dst','standalone_gpu','cores',1,'mwa',2);
Everything can of course also be set up through the alignment project GUI. The numerical parameters would correspond to:
Check, unfold and run the project through the alignment project GUI or through the command line:
dvcheck aliVLP dvunfold aliVLP dvrun aliVLP
On one GPU and 2 cores the alignment project should not take longer than half an hour. Check the status of the alignment project with
dvstatus aliVLP
You can also see the average of the last computed iteration in dmapview with the command. If needed, adjust the lowpass and contrast to improve the depiction.
ddb aliVLP:a -m
Recenter and recrop
After the alignment project finished successfully we can inspect the last computed average with the same command introduced before:
ddb aliVLP:a -m
The command can be extended to see the averages of all iterations including the starting reference. Once in dmapview, press the button norm to properly compare the averages.
ddb aliVLP:a:ite=0:last -m
In the last average, you should already start to see a C6 symmetry. However, the symmetry axis is most probably not centered yet. We now want to realign the particles such that their symmetry axis is parallel to the Z axis. We also want to make sure that the particles are centered in the middle of the subvolumes. We do all this in two steps. First we subbox the particles using a newly defined center and recrop the particles with a smaller boxsize. Second we align the axis of symmetry using a synthetic template. Both procedures are described in detail in the advanced starters guide in the section Aligning the axis of symmetry and Subboxing and are now applied here.
Step 1: Subboxing
Open the last computed average in dmapview as previously described. Under Clicks activated the radio buttons for the center/north tool. Find the coordinates of the center of one subunit of the lattice. In our case we found the center of a subunit at [88,75,72]. For you these coordinates might be different. The center of a subvolume with a sidelength of 128 pixels is defined as [65,65,65]. We use the found coordinates to compute the vector from the center of the subvolume to the center of a subunit.
rSubunitFromCenter = [88,75,72] - [65,65,65];
This shift will now be applied to all particles in the table. Read the last computed table of the alignment project:
ddb aliVLP:rt -ws t
Apply the new center to the table
ts = dynamo_subboxing_table(t,rSubunitFromCenter);
The table now contains the coordinates of the recentered particles. We use this new table to recrop the particles. We first define a new target folder:
targetFolder = './particlesSize96r';
Then we recrop the new particles. Since the particles are centered now, we can also choose a smaller boxsize of 96 pixels. This will save space and speed up the processes.
dtcrop('VLPtomograms.doc',ts,targetFolder,96);
Again we average and visualize the cropping results:
finalTable = dread([targetFolder,'/crop.tbl']); oa = daverage(targetFolder,'t',finalTable,'fc',1); dwrite(oa.average,[targetFolder '/template.em']);
Step 2: Align symmetry axis
We see that the new average is niceley centered (there is one subunit of the lattice in the center of the volume). However, the symmetry axis is not aligned with the Z axis (the whole average looks a bit 'tilted'). We can fix that by aligning the last computed average to a synthetic template and then apply the found transformation to the table, resulting in a new and correct orientation for all particles.
First we create a synthetic template using the mask tools of Dynamo. We choose a curved membrane with a hole in the center, representing the 'hollow' central subunit of the lattice.
mr = dpktomo.examples.motiveTypes.Membrane(); % create membrane object mr.thickness = 22; % choose thickness of membrane mr.sidelength = 96; % choose sidelength of box mr.getMask(); % compute mask mem = (mr.mask)*(-1)+1; % invert contrast cyl = dynamo_cylinder(7,96,[48,48,48]); % create a cylinder (this will be the 'hole') templateSum = mem+cyl; % sum the two masks template = templateSum; template(template>0) = 1; % binarize the new mask
We look at the newly created template to make sure it was computed correctly.
dview(template)
Now we align the last computed average of the previous alignment project to the newly created template. We save the transformation parameters in the object sal.
sal = dalign(oa.average,template,'cr',30,'cs',5,'ir',0,'dim',48,'limm',1,'lim',[15,15,15]);
A quick look at the newly aligned average should show a nicely centered particle with the symmetry axis aligned to the Z axis.
dmapview(sal.aligned_particle);
The transformation parameters that have been saved in the object sal are now applied to the table
tr = dynamo_table_rigid(finalTable,sal.Tp);
We now have an up to date table with nicely centered and aligned particles. This new table can be used in a new, finer alignment project.
Volume exclusion
Before starting the new alignment project we apply one more operation on the table. Since we
% volume filter:
trEx = dpktbl.exclusionPerVolume(tr,20);
Second aligment project
Use C6 symmetriy in the alignment projects finer parameters tighter mask
% create a tighter mask using a thick membrane mr = dpktomo.examples.motiveTypes.Membrane(); mr.thickness = 55; mr.sidelength = 96; mr.getMask(); mem_mask = mr.mask;
dwrite(mem_mask,'mem_mask_thick.em')
check in dmapview that mask is good
dmapview(oa.average)
and then in mask put path of mask and click layer.
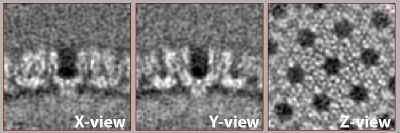
Result
After the second alignment and without any postprocessing or elaborate refinement your final average may look similar to this one. If not it may need one more round with finer parameters, tighter mask or one more step of recentering and recropping. This representation was lowpass filtered to ~8 angstroms.